Resources
News & Events
Upcoming USDM events, partner sessions, and field briefings for regulated life sciences teams.
Featured events
Showing 3 events

VeevaR&DQualityVault
USDM at Veeva R&D and Quality Summit 2026
USDM is sponsoring Veeva R&D and Quality Summit 2026, where life sciences R&D, Quality, Regulatory, Safety, Clinical, and IT leaders gather to advance Veeva programs and regulated operations.
October 20–21, 2026Boston, MA
View event
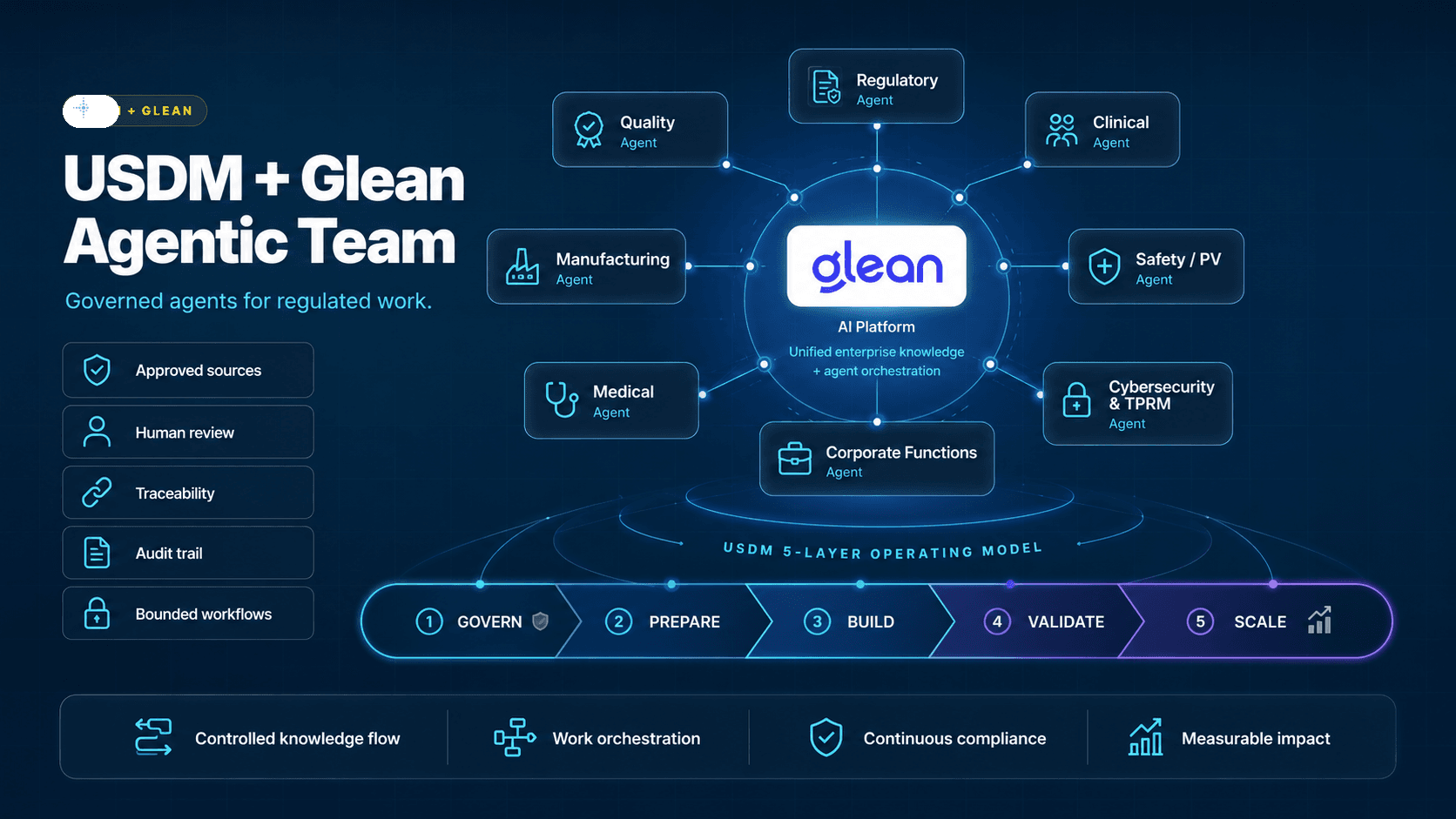
GleanEnterprise AIWork AILife sciences
USDM at Glean:GO 2026
USDM is sponsoring Glean:GO 2026, Glean’s flagship enterprise AI conference for leaders building the next generation of AI-powered work.
August 26–27, 2026Fort Mason, Festival and Gateway Pavilion — San Francisco, CA
View event

ServiceNowLife sciencesGxPAI workflows
ServiceNow Knowledge26 – Life Sciences Lunch & Learn
Join USDM, ServiceNow’s exclusive Life Sciences partner, for an intimate lunch session on deploying AI-powered workflows across the enterprise without sacrificing GxP integrity.
May 7, 2026 | 12:30 – 2:00 PMMercato Della Pescheria, St. Mark’s Square, The Venetian — Las Vegas, NV
View event