Add Your Heading Text Here
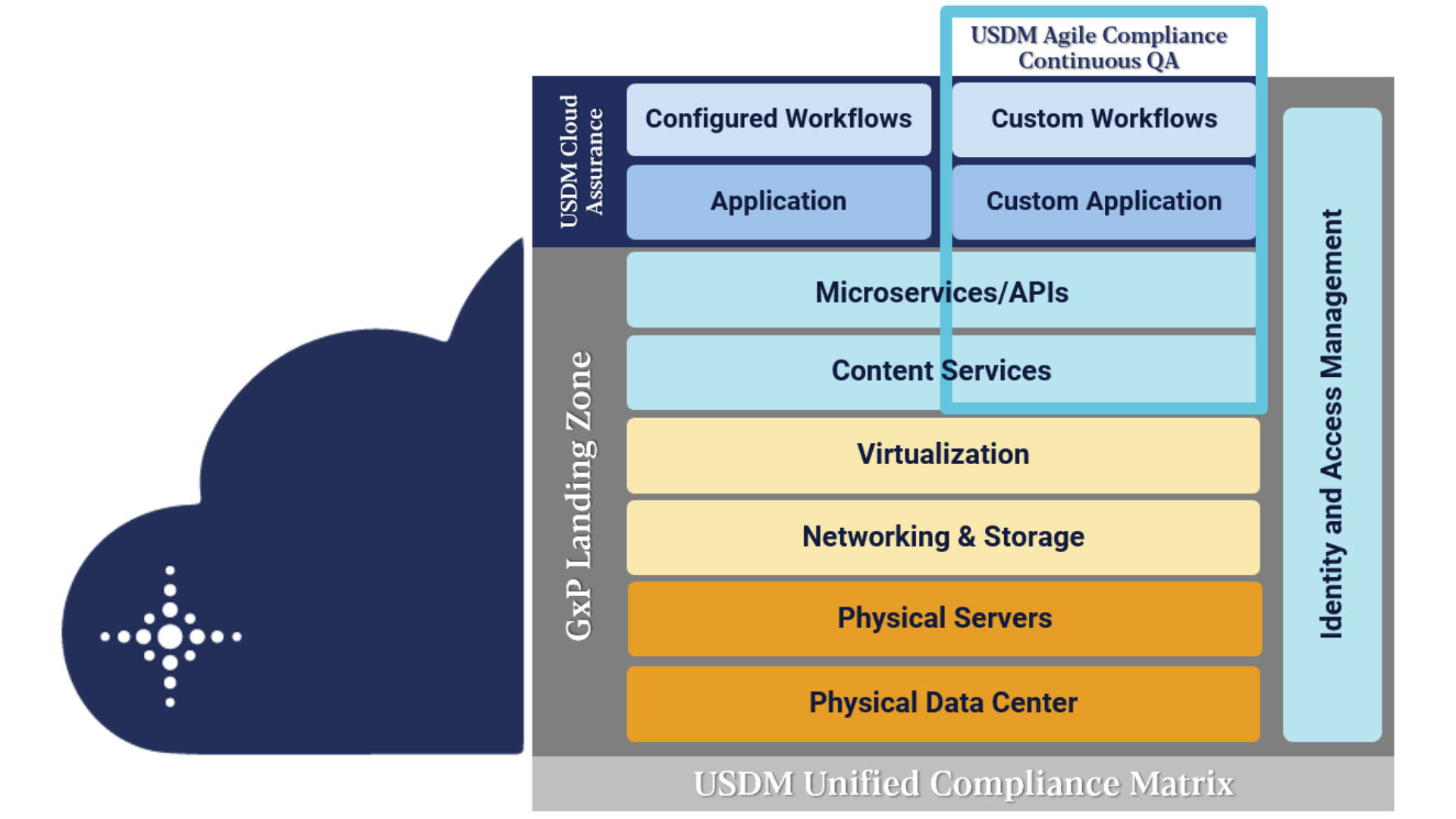
1) GxP Landing Zone
Boldly reimagine your GxP business requirements by harmonizing your Google Cloud stack for regulated and non-regulated use. USDM has created a GxP landing zone, purpose-built for the life sciences and designed to 21 CFR Part 11, Annex 11, and relevant GxP requirements providing a pre-configured environment – provisioned through code – to host GxP workloads in the cloud.
Google Cloud has unmatched data processing power to bring your life-saving therapies and drugs to market faster. USDM has the strategy and execution know-how to keep you in continuous compliance and relieve your validation burdens.
Download our datasheet to learn more >>>
USDM is proud to be a Google Cloud Partner

Watch this video to learn how USDM delivers a GxP Landing Zone for Google Cloud in 8-9 weeks.
2) USDM's Vendor Assurance Report
Google Cloud achieved its Cloud Assurance Certification by passing USDM’s rigorous assessment that satisfies 34 worldwide health authorities and 6,500+ regulatory requirements globally. We have published the results of our Google Cloud assessment in our Vendor Assurance Report. You can access and leverage this report as regulatory evidence, which minimizes your internal validation effort.
To request a copy, please fill out this form. >>>
USDM’s Cloud Assurance Certification recognizes technology vendors that meet the quality and compliance demands of the life sciences industry and is THE badge of trust for GxP-ready functionality. Learn more about our certification here.
Request a copy of our Vendor Assurance Report for Google Cloud

3) Continuous Compliance
USDM’s Cloud Assurance service delivers end-to-end GxP compliance, including rapid implementation, qualification, and release management maintenance to enable a continuously compliant tech stack.
To reap the greatest benefits of the cloud, you need to think holistically about your tech stack. Getting your infrastructure to the cloud provides you with faster delivery, lower cost to manage compliance, and greater efficiency. We use a flexible cloud adoption model to migrate regulated workloads to Google Cloud based on your IT and quality priorities.
To learn more about Cloud Assurance for Google Cloud, read this white paper: Google Cloud Platform for Life Sciences and Health Technology.
